Research collaboration between FDA and Owlstone Medical set to see improvements in breath biopsies
Owlstone Medical has entered into a research collaboration with the US Food and Drug Administration’s (FDA’s) Centre for Devices and Radiological Health to develop methods for the confident identification of individual chemicals in the composure of breath.
The goal of the collaboration is to develop methods for the conf ident identification of individual chemicals in breath, according to the press release.
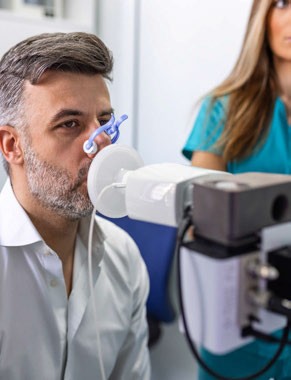
Owlstone Medical will use the outcomes from the collaboration to enable in vitro diagnostic device manufacturers, academic researchers, government researchers and biopharma to identify breath biomarkers related to disease with confidence.
In addition to its work with the Bill and Melinda Gates Foundation, Owlstone Medical will beutilising the research from this five-year collaboration to support further investigation into the field of identifying disease biomarkers, in addition to further developing portable breath diagnostic devices.
Billy Boyle, co-founder and CEO at Owlstone Medical, said: “By defining rigorous practices for analysis and harnessing the collective knowledge and experience of the FDA, we aim to collaboratively create a common set of analytical methodologies that will serve as a valuable reference for the entire breath research community. We believe these regulatory science tools (RSTs) have the potential to accelerate the future development of simple and low-cost, non-invasive diagnostic tests and portable devices. We are proud and delighted to have been chosen as their partner in this important project.”
The collaboration aims to develop RSTs for the identification of volatile organic compounds (VOCs) in complex mixtures.
Owlstone Medical is a biotech company based in the UK.