R & D
Pharmaceutical research and development updates
Articles
Medincell and AbbVie have announced that they have entered into a collaboration to co-develop and commercialise up to six next-generation long-acting injectables (LAI) across multiple therapeutic areas and indications.

UCB has announced that the FDA has accepted for review its supplemental biologics license application (sbla) for bimzelx (bimekizumab-bkzx), for the treatment of adult patients with moderate-to-severe hidradenitis suppurativa (hs).
Sanofi has announced positive results from its phase 3 LUNA 3 study, which assessed Rilzabrutinib 400mg twice daily oral treatment in adult patients with persistent or chronic immune thrombocytopaenia (itp).
GSK has announced positive results from its phase 3 EAGLE-1 trial for Gepotidacin, a potential first-in-class oral antibiotic for the treatment of uncomplicated urogenital gonorrhoea (GC) in adolescent and adult patients.

Novartis has announced new results from a pre-specified interim analysis of its phase 3 APPLAUSE-igan trial of Fabhalta (iptacopan),an investigational factor b inhibitor of the alternative complement pathway, in patients with iga nephropathy (igan).
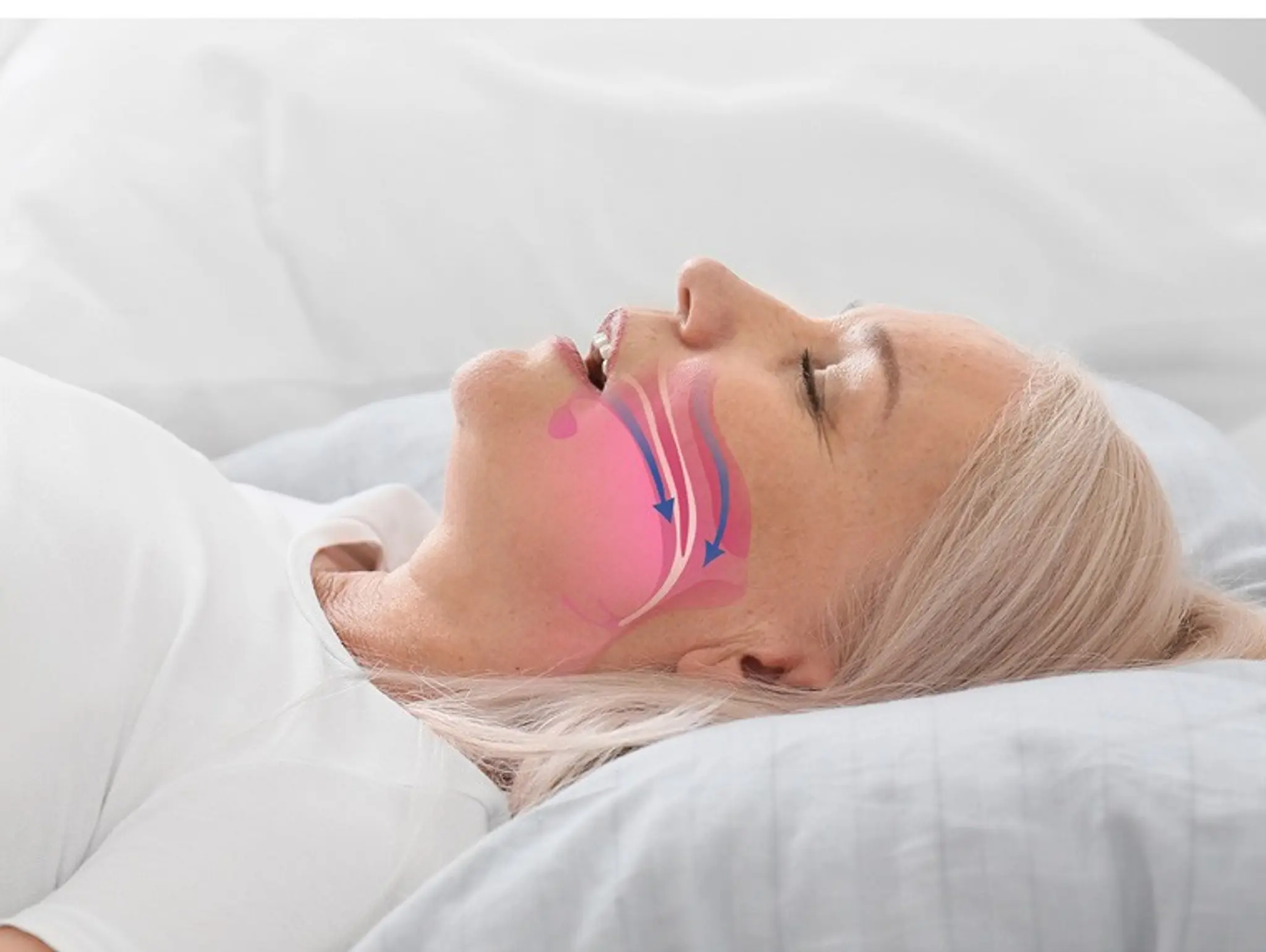
Eli Lilly has announced positive topline results from its SURMOUNT-osa phase 3 trials, which demonstrated that tirzepatide injection (10mg or 15mg) significantly reduced the apnoea-hypopnea index (ahi) compared to placebo, therefore meeting its primary endpoints.

Neurolentech has announced that it has entered into a technology access partnership with Kaerus Bioscience, with the aim of driving advancement in neurodevelopmental disorder research.

Ardena has announced that it has received good manufacturing practice (GMP) approval from the Dutch Healthcare Authority for its new analytical laboratories in its expanded facility.

Swedish biotech startup Ironic Biotech has announced that it has successfully closed a pre-seed funding round of $1m, led by Nordic Foodtech VC. The company intends to use this funding to produce a solution to the global issue of iron deficiency and anaemia.

Curve Therapeutics has announced that its chief scientific officer, professor Ali Tavassoli, has published research in the Journal of the American Chemical Society (JACS), assessing dual hypoxia inducible factors (hif)-1 and -2 inhibitors in cancer treatment.
Gilead sciences and Merck (known as MSD outside of the US and Canada) have announced results from the phase 2 clinical study assessing the combination of islatravir and lenacapavir for viral suppression in HIV treatment.
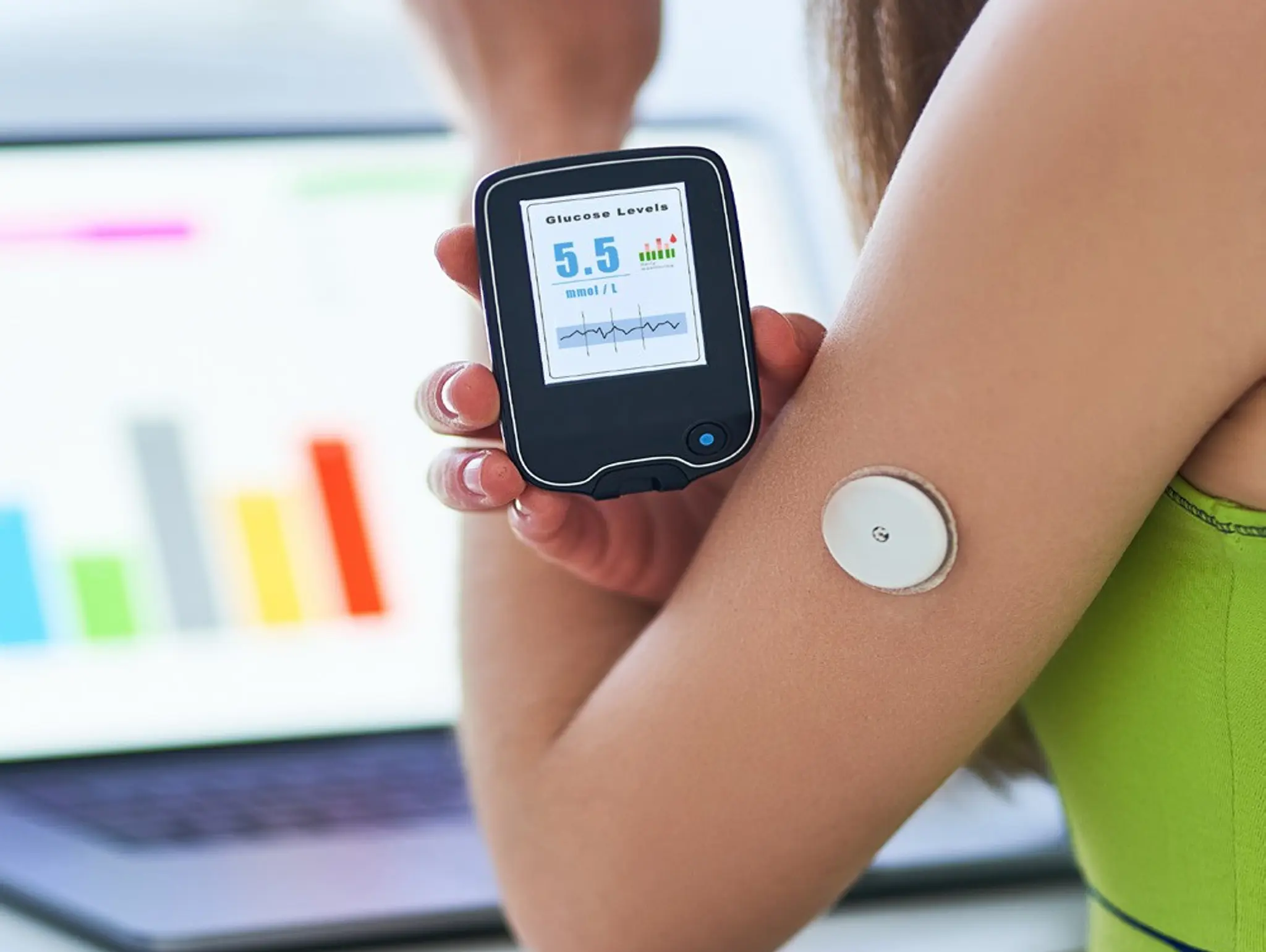
Roche has shared the latest data on its novel solution for continuous glucose monitoring (CGM), including a CGM sensor and two apps designed to present current glucose values and predictions over 30 minutes and two hours.