
Global
The latest news from the global pharmaceutical industry
Articles

Eko Health has announced that the FDA has approved its low ejection fraction (low ef) detection artificial intelligence (AI).
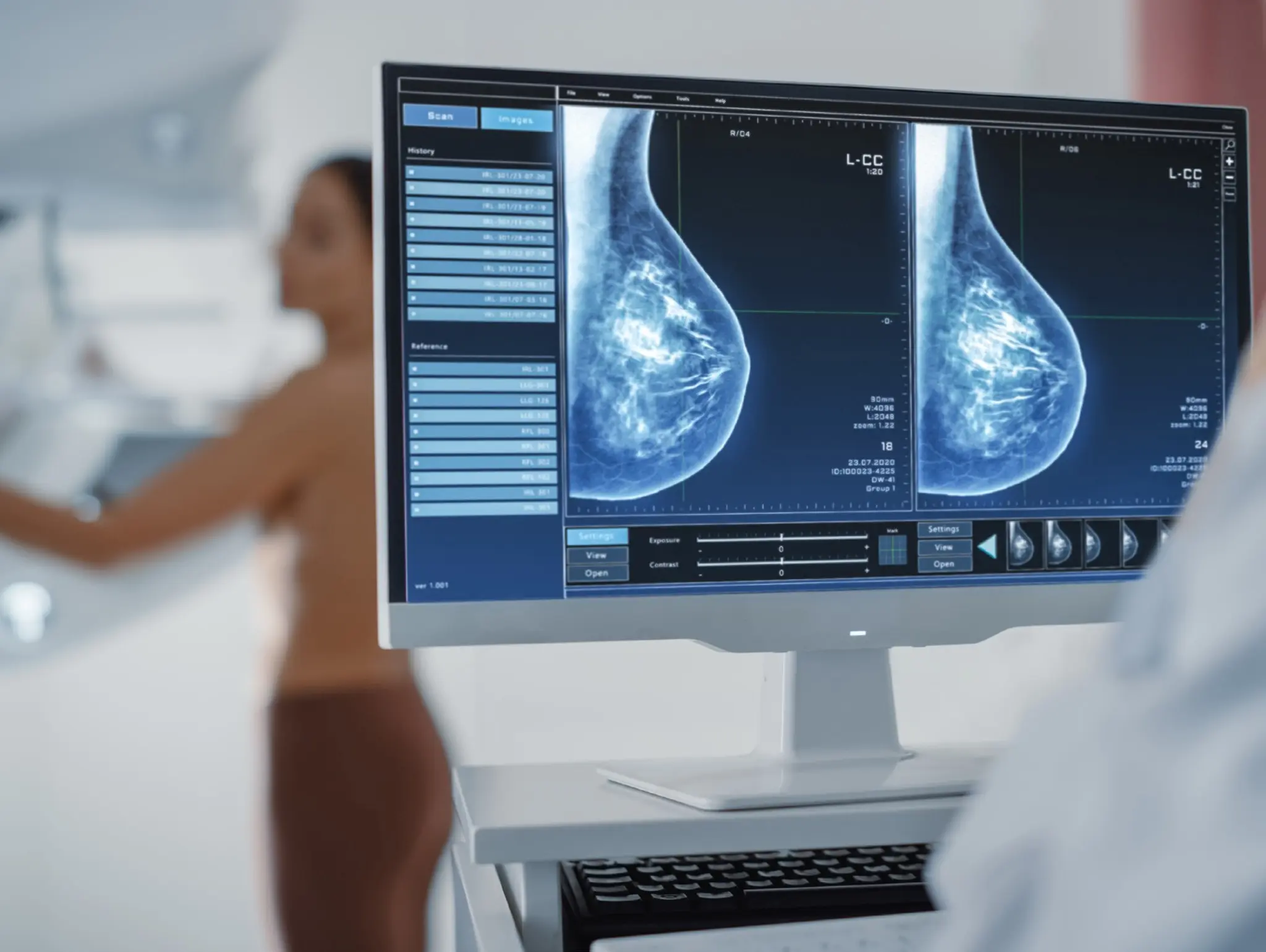
AstraZeneca and Daiichi Sankyo have announced that their biologics license application (bla) for datopotamab deruxtecan (dato-dxd) has been accepted in the US for the treatment of adult patients with unresectable or metastatic hormone receptor (hr)-positive, her2-negative breast cancer who have had prior systemic therapy for unresectable or metastatic disease.
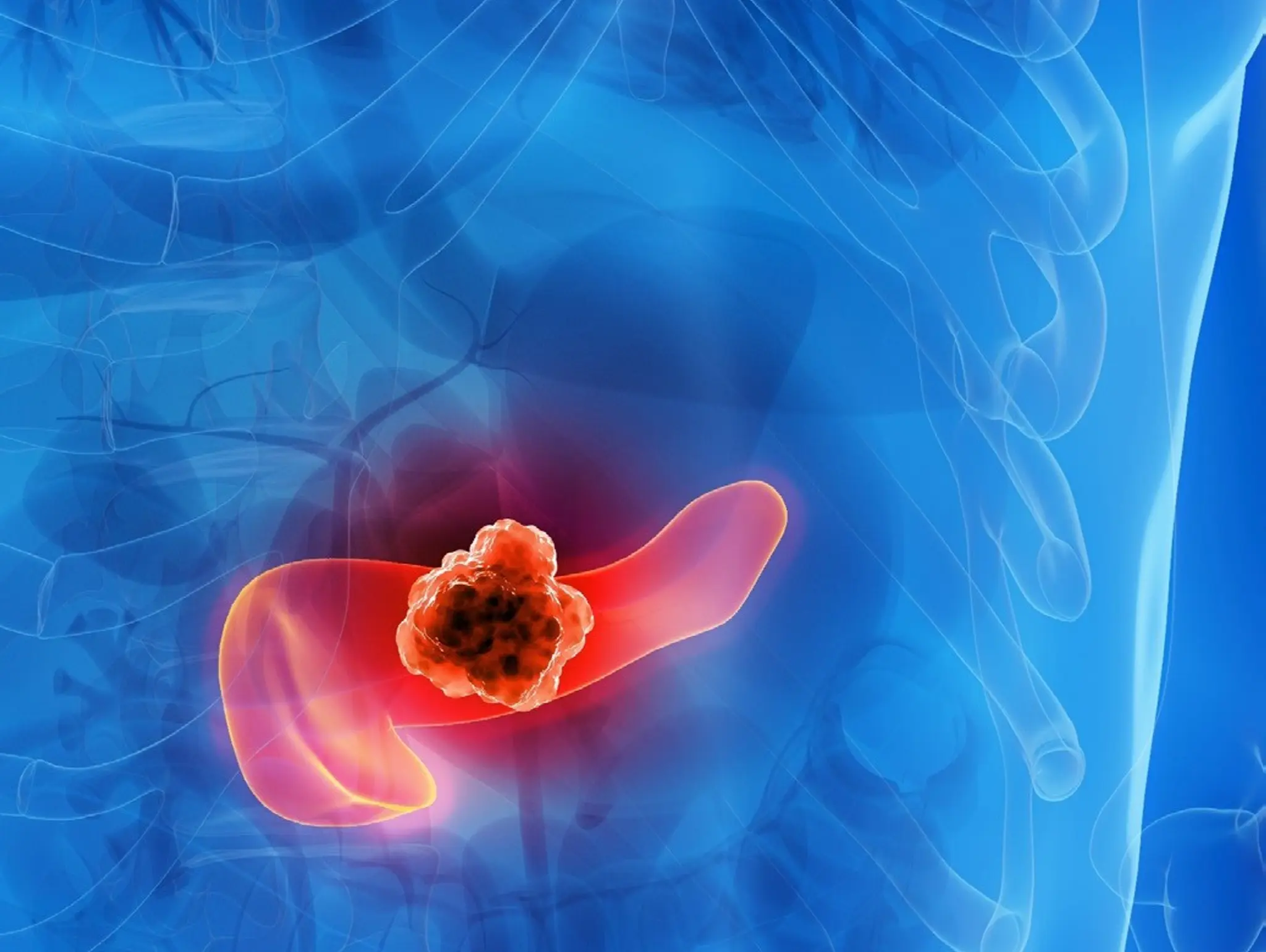
Candel Therapeutics has announced that the FDA has granted orphan drug designation (ODD) to can-2409, Candel’s immunotherapy candidate, for the treatment of pancreatic cancer.
GSK has announced that the FDA has accepted for review a biologics license application (BLA) for its 5-in-1 meningococcal abcwy (menabcwy) vaccine candidate.
Roche has announced that the FDA has approved alecensa (alectinib) for the adjuvant treatment following tumour resection for patients with anaplastic lymphoma kinase (alk)-positive non-small cell lung cancer (nsclc).

ImmunityBio has announced that the FDA has approved Anktiva (n-803, or nogapendekin alfa inbakicept-pmln) plus bacillus calmette-guérin (bcg) for the treatment of patients with bcg-unresponsive non-muscle invasive bladder cancer (nmibc) with carcinoma in situ (cis), with or without papillary tumours.
The FDA has announced that it has approved Rezdiffra (resmetirom) for the treatment of adult patients with noncirrhotic non-alcoholic steatohepatitis (NASH) with moderate-to-advanced liver scarring (fibrosis), for use alongside diet and exercise.
Spero Therapeutics has announced that it has gained FDA clearance for the investigational new drug (IND) application to assess SPR206 in a phase 2 clinical study.
Johnson & Johnson (J&J) has announced that the FDA has approved Opsynvi (macitentan and tadalafil) for the chronic treatment of adults with pulmonary arterial hypertension (PAH).
AstraZeneca has announced that the FDA has approved Ultomiris (ravulizumab-cwvs) as the first and only long-acting c5 complement inhibitor for the treatment of adult patients with anti-aquaporin-4 (aqp4) antibody-positive (ab+) neuromyelitis optica spectrum disorder (nmosd).
AstraZeneca has announced that the FDA has approved Tagrisso for the treatment of adult patients with locally advanced or metastatic epidermal growth factor receptor-mutated non-small cell lung cancer.
GSK has announced that the FDA has granted fast track designation for bepirovirsen, an investigational antisense oligonucleotide for the treatment of chronic hepatitis b.